Intercellular Communication Breakdown in Aging
Explore how the breakdown of intercellular communication, a hallmark of aging, contributes to age-related decline and disease, and potential interventions.
Table of Contents
DISCLAIMER
This article is for informational purposes only and does not constitute medical advice. The statements in this article have not been evaluated by the FDA. The information presented is based on published research and should not be used as a substitute for professional medical guidance. Consult your physician before starting any supplement or health protocol.
Our bodies are marvels of biological engineering, composed of trillions of cells, each with specialized functions. Yet, these cells do not operate in isolation. They form a vast, intricate network, constantly exchanging information to coordinate their activities, maintain tissue integrity, and ensure the smooth functioning of organs and systems. This complex dialogue, known as intercellular communication, is fundamental to life itself.
However, as we age, this vital communication network appears to falter. Research suggests that a progressive breakdown in the ability of cells to effectively send, receive, and interpret signals is a significant, yet often overlooked, hallmark of aging. This dysregulation in cellular crosstalk contributes to the decline in tissue function, impaired repair mechanisms, and increased susceptibility to age-related diseases. Understanding the mechanisms behind this communication breakdown offers a compelling avenue for developing interventions aimed at promoting healthier, longer lives.
What is Intercellular Communication and Why is it Essential?
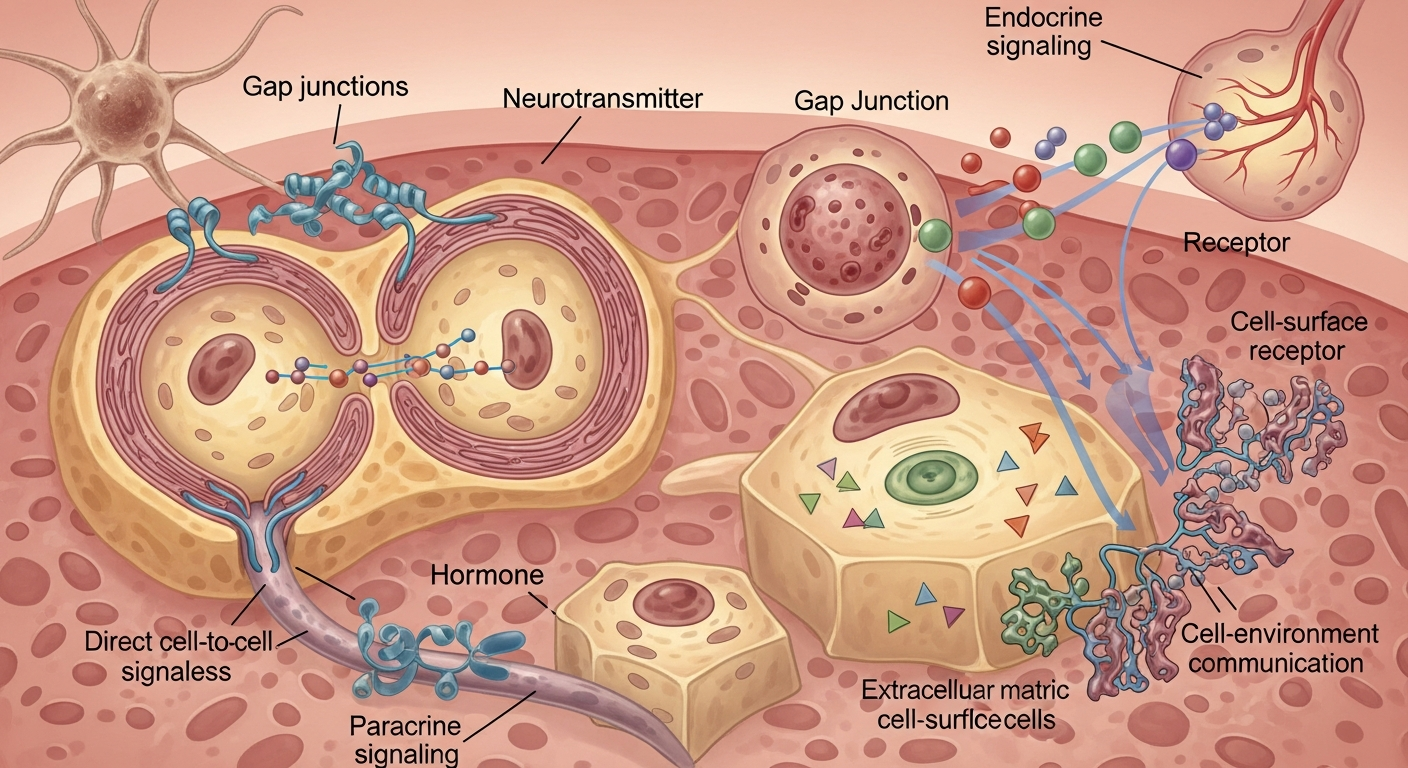
At its core, intercellular communication is the process by which cells interact with each other and their environment. It’s the cellular language that allows for the precise orchestration of biological processes, from embryonic development to immune responses and metabolic regulation. Without effective communication, a multicellular organism could not function cohesively.
What are the Different Modes of Cellular Crosstalk?
Cells employ various sophisticated mechanisms to communicate, each suited for different distances and types of messages:
- Paracrine Signaling: Cells release signaling molecules that act on nearby target cells. A classic example is the release of growth factors that stimulate the proliferation of adjacent cells.
- Autocrine Signaling: A cell releases signaling molecules that then bind to receptors on its own surface, affecting its own behavior. This is often seen in immune cells and cancer cells.
- Endocrine Signaling: Cells release hormones into the bloodstream, which then travel to distant target cells throughout the body. The endocrine system, involving hormones like insulin or thyroid hormones, is a prime example.
- Juxtacrine Signaling (Direct Contact): Cells communicate through direct physical contact, either via gap junctions (small channels connecting cytoplasm) or through surface-bound proteins that interact.
- Synaptic Signaling: A specialized form of paracrine signaling occurring in the nervous system, where neurons transmit signals rapidly across a synapse using neurotransmitters.
- Extracellular Vesicles (EVs): Cells release tiny membrane-bound sacs (like exosomes and microvesicles) containing proteins, lipids, and nucleic acids (e.g., microRNAs) that can be taken up by distant cells, delivering complex messages.
These modes of communication rely on a diverse array of messengers, including hormones, growth factors, cytokines, neurotransmitters, chemokines, and even mechanical forces transmitted through the extracellular matrix (ECM). The integrity and efficiency of these communication pathways are paramount for maintaining cellular and tissue homeostasis.
How Does Cellular Communication Change with Age?
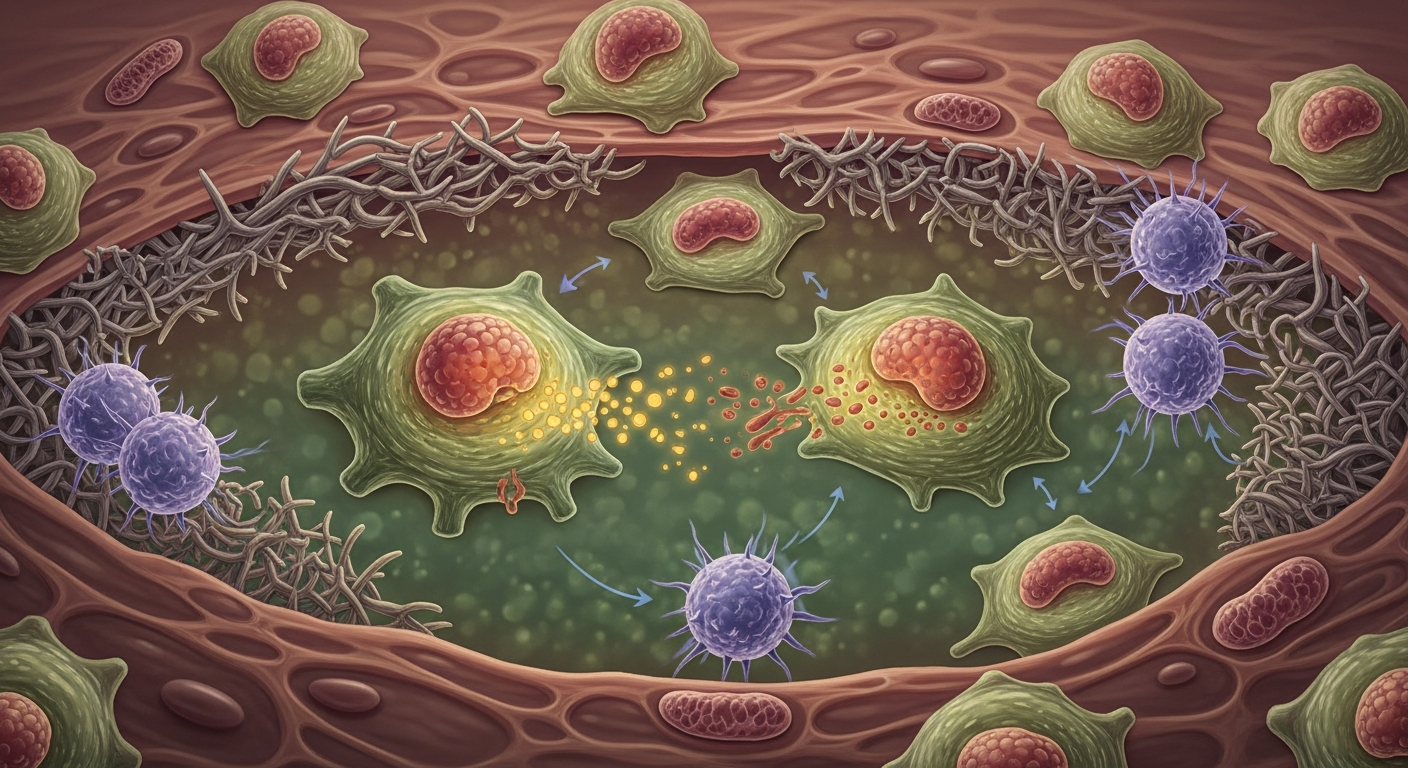
The aging process appears to profoundly impact the delicate balance of intercellular communication, leading to a state of chronic dysregulation that contributes to functional decline. This breakdown is not a single event but rather a multifaceted process involving alterations at various levels of cellular interaction.
Does Aging Dysregulate Key Signaling Pathways?
One of the most well-documented aspects of aging’s impact on intercellular communication involves the dysregulation of fundamental signaling pathways that control cell growth, metabolism, and stress responses.
Insulin/IGF-1 Signaling (IIS)
The insulin/insulin-like growth factor 1 (IGF-1) signaling pathway is a conserved regulator of metabolism and lifespan across many species. Research suggests that while acute activation of IIS is vital for growth and energy metabolism, chronic over-activation or dysregulation of this pathway, particularly in later life, appears to contribute to aging processes (Kenyon, 2010; PMID: 20498701).
With age, many tissues may develop a form of “insulin resistance,” where cells become less responsive to insulin signals, leading to higher circulating insulin levels. Similarly, changes in IGF-1 sensitivity can occur. This dysregulation can impair nutrient sensing, promote anabolic processes over repair, and contribute to metabolic disorders common in aging. Studies indicate that reducing IIS activity, often through genetic or pharmacological interventions, can extend lifespan in various model organisms (Fontana et al., 2014; PMID: 24796340).
mTOR Pathway (Mechanistic Target of Rapamycin)
The mTOR pathway is another central nutrient-sensing pathway that integrates signals from growth factors, energy status, and amino acids to regulate cell growth, proliferation, and protein synthesis. While essential for these processes, chronic activation of mTOR has been linked to accelerated aging in mammals.
As we age, there appears to be an increase in basal mTOR activity in some tissues, potentially driven by persistent nutrient signals or inflammatory cues. This sustained activation may suppress autophagy (the cellular recycling process), promote cellular senescence, and contribute to age-related pathologies. Inhibiting mTOR activity, for instance with drugs like rapamycin, has shown promise in extending lifespan and healthspan in various animal models, suggesting that modulating this pathway can restore a more youthful cellular communication balance (Johnson et al., 2019; PMID: 31109918).
AMPK Pathway (AMP-activated Protein Kinase)
AMPK is a crucial energy sensor that is activated when cellular energy levels are low (high AMP:ATP ratio). Its activation promotes catabolic processes that generate ATP (like fatty acid oxidation and glucose uptake) and inhibits anabolic processes that consume ATP (like protein synthesis and lipid synthesis).
In aging, there appears to be a decline in AMPK activity in many tissues. This reduction in the “energy sensor” can lead to impaired metabolic flexibility, reduced mitochondrial function, and an imbalance between energy production and consumption. A less active AMPK pathway may also contribute to the chronic activation of mTOR, further exacerbating age-related cellular dysfunction. Interventions that activate AMPK, such as exercise and certain drugs like metformin, are being investigated for their potential anti-aging effects by restoring metabolic signaling and cellular communication (Salminen & Kaarniranta, 2012; PMID: 22806297).
How Does the Extracellular Matrix Contribute to Communication Breakdown?
The extracellular matrix (ECM) is not merely a structural scaffold; it is a dynamic biochemical and biomechanical signaling hub that profoundly influences cell behavior and communication. Composed of proteins like collagen, elastin, and fibronectin, along with proteoglycans and glycosaminoglycans, the ECM provides physical support, sequesters growth factors, and transmits mechanical cues.
Stiffening and Altered Composition
With advancing age, the ECM undergoes significant changes. There is a general increase in stiffness, primarily due to the accumulation of advanced glycation end products (AGEs) that cross-link collagen fibers, making tissues less elastic and more rigid. Elastin fibers, which provide elasticity, also degrade and become less functional (Ricard-Blum & Ruggiero, 2016; PMID: 26692994).
This altered ECM composition and mechanics directly impact intercellular communication:
- Impaired Mechanotransduction: Cells sense and respond to the physical properties of their environment. A stiffened ECM can alter gene expression, cell differentiation, and migration, pushing cells towards a senescent or pro-fibrotic state.
- Reduced Growth Factor Availability: The ECM often acts as a reservoir for growth factors and cytokines. Age-related changes can alter the binding and release of these signaling molecules, making them less available or less effectively presented to cells.
- Barrier to Diffusion: A denser, more cross-linked ECM can impede the diffusion of nutrients, oxygen, and signaling molecules, creating microenvironments where cells are less able to communicate or receive vital resources.
These ECM changes can affect various tissues, including skin, blood vessels, and cartilage, contributing to impaired wound healing, cardiovascular stiffening, and joint degeneration.
What Role Do Senescent Cells and the SASP Play?
Cellular senescence is a state of irreversible cell cycle arrest that cells enter in response to various stressors, including telomere shortening, DNA damage, and oncogenic stress. While senescent cells initially play protective roles (e.g., in wound healing or tumor suppression), their accumulation with age is strongly implicated in tissue dysfunction and disease.
The Senescence-Associated Secretory Phenotype (SASP)
A critical aspect of senescent cells’ impact on intercellular communication is their acquisition of a senescence-associated secretory phenotype (SASP). Senescent cells secrete a potent cocktail of pro-inflammatory cytokines (e.g., IL-6, IL-8), chemokines, growth factors, and matrix-degrading proteases (e.g., MMPs) (Coppé et al., 2010; PMID: 20164448).
The SASP acts as a powerful disruptor of local and systemic intercellular communication:
- Propagation of Senescence: SASP factors can induce senescence in neighboring healthy cells, creating a self-perpetuating cycle of cellular aging.
- Chronic Inflammation: The pro-inflammatory components of the SASP contribute to “inflammaging,” a chronic low-grade inflammation that characterizes aging and underlies many age-related diseases.
- Impaired Tissue Regeneration: SASP factors can impair the function of stem cells and progenitor cells, hindering the body’s ability to repair and regenerate damaged tissues.
- ECM Remodeling: SASP-secreted MMPs can degrade the ECM, but this degradation is often dysregulated, contributing to abnormal tissue architecture rather than healthy remodeling.
The accumulation of senescent cells and their SASP represents a significant burden on the body’s communication systems, creating a toxic microenvironment that drives dysfunction in surrounding healthy cells (Gorgoulis et al., 2019; PMID: 31346294).
Are Extracellular Vesicles Affected by Aging?
Extracellular vesicles (EVs), including exosomes and microvesicles, are tiny lipid-bound sacs released by cells that carry a diverse cargo of proteins, lipids, and nucleic acids (especially microRNAs). They act as important mediators of long-distance intercellular communication, enabling cells to exchange complex information without direct contact.
Altered Cargo and Reduced Efficiency
Research indicates that EV-mediated communication is altered with age. Senescent cells, for instance, release EVs with a distinct cargo that can promote senescence or inflammation in recipient cells (Xu et al., 2018; PMID: 30287103). Furthermore, the quantity and quality of EVs released by healthy cells may also change, potentially affecting their ability to deliver beneficial signals.
These age-related changes in EVs can disrupt various processes:
- Immune Modulation: EVs play a role in immune regulation. Age-related changes in immune cell EVs might contribute to immune dysfunction.
- Tissue Repair: EVs from young, healthy cells can promote tissue repair and regeneration. If aged cells release less effective or even detrimental EVs, this could impair regenerative capacity.
- Neurodegenerative Processes: EVs are involved in neuron-to-neuron communication and the spread of misfolded proteins in neurodegenerative diseases. Age-related EV dysregulation may contribute to these pathologies.
The precise impact of aging on EV communication is an active area of research, but evidence suggests it is a crucial component of the overall communication breakdown.
Do Gap Junctions and Cell Adhesion Decline with Age?
Direct cell-to-cell communication and adhesion are vital for maintaining tissue structure and function. Gap junctions are specialized intercellular channels that allow for the direct passage of small molecules and ions between adjacent cells, facilitating rapid and coordinated responses. Cell adhesion molecules mediate physical connections between cells and between cells and the ECM.
Reduced Connexin Expression and Function
Studies suggest that the expression and function of connexins, the proteins that form gap junctions, can decline with age in various tissues. For example, reduced gap junction coupling has been observed in aging heart muscle, potentially contributing to cardiac arrhythmias (Jansen et al., 2010; PMID: 20485600). Similarly, changes in cell adhesion molecules can weaken tissue integrity and alter cellular responses to mechanical cues.
This impairment in direct contact communication can lead to:
- Loss of Tissue Cohesion: Weaker cell-cell and cell-ECM adhesion can compromise tissue integrity, making tissues more fragile and susceptible to damage.
- Dysregulated Cell Growth and Differentiation: Gap junctions play roles in controlling cell proliferation and differentiation. Their decline might contribute to uncontrolled cell growth or impaired regenerative processes.
- Impaired Metabolic Coupling: In some tissues, gap junctions allow for metabolic cooperation between cells. Their dysfunction could lead to metabolic stress in specific cell populations.
Is Neuroendocrine Dysregulation a Factor?
The neuroendocrine system, a complex interplay between the nervous and endocrine systems, serves as a master regulator of systemic communication. Hormones released by endocrine glands and neurotransmitters released by neurons coordinate physiological responses across the entire body.
Hormonal Decline and Receptor Sensitivity
Aging is associated with a decline in the production of many hormones, such as sex hormones (estrogen, testosterone), growth hormone, and dehydroepiandrosterone (DHEA). Furthermore, target tissues may become less responsive to existing hormones due to changes in receptor expression or post-receptor signaling (Finch & Cohen, 2017; PMID: 28254425).
This neuroendocrine dysregulation contributes to widespread communication breakdown:
- Metabolic Syndrome: Hormonal imbalances contribute to age-related changes in glucose metabolism, lipid profiles, and body composition.
- Immune System Decline: Hormones like corticosteroids and sex steroids modulate immune responses. Their age-related changes contribute to immunosenescence.
- Cognitive Decline: Hormones play critical roles in brain function. Their decline can impact neuronal communication and contribute to cognitive impairment.
- Sleep Disturbances: Melatonin, a hormone regulating circadian rhythms, often declines with age, contributing to sleep disruptions.
The hypothalamic-pituitary-adrenal (HPA) axis, which regulates stress responses, also shows age-related changes, potentially leading to chronic stress states that further impair cellular communication throughout the body.
How Does the Immune System Contribute to Communication Breakdown?
The immune system is a sophisticated communication network itself, constantly surveying the body for threats and coordinating responses. However, with age, the immune system undergoes significant changes, often termed “immunosenescence,” which contributes to a state of chronic low-grade inflammation known as “inflammaging.”
Chronic Low-Grade Inflammation (Inflammaging)
Inflammaging is characterized by elevated levels of pro-inflammatory cytokines (e.g., TNF-α, IL-6, IL-1β) in the absence of overt infection. This persistent inflammatory state is driven by various factors, including the accumulation of senescent cells and their SASP, dysfunctional mitochondria, and persistent infections.
Inflammaging profoundly disrupts intercellular communication:
- Altered Signaling: Chronic exposure to inflammatory cytokines can desensitize cells to beneficial signals and promote pathological responses.
- Tissue Damage: Persistent inflammation can directly damage tissues and impair their regenerative capacity, creating a vicious cycle.
- Impaired Immune Surveillance: The aged immune system is less effective at clearing pathogens and cancerous cells, partly due to altered communication between immune cells themselves.
- Systemic Effects: Inflammatory mediators can travel throughout the body, affecting distant organs and contributing to conditions like atherosclerosis, neurodegeneration, and sarcopenia (Franceschi et al., 2013; PMID: 24021798).
The immune system, therefore, not only experiences communication breakdown internally but also actively contributes to the disruption of communication in other tissues through its pro-inflammatory output.
Consequences of Impaired Intercellular Communication in Aging
The multifaceted breakdown of intercellular communication appears to have wide-ranging consequences for health and longevity, contributing to many of the observable phenotypes of aging and increasing susceptibility to age-related diseases.
Table 1: Key Intercellular Communication Mechanisms and Age-Related Changes
| Communication Mechanism | Primary Messengers/Components | Age-Related Change | Potential Consequences for Aging |
|---|---|---|---|
| Signaling Pathways | Insulin, IGF-1, Nutrients, Growth Factors | Dysregulation (e.g., IIS resistance, chronic mTOR, reduced AMPK) | Metabolic dysfunction, reduced autophagy, impaired stress response, accelerated cellular aging |
| Extracellular Matrix (ECM) | Collagen, Elastin, Proteoglycans, Mechanical Cues | Increased stiffness, cross-linking, altered composition | Impaired mechanotransduction, reduced nutrient diffusion, poor tissue repair, organ fibrosis |
| Senescent Cells (SASP) | Pro-inflammatory cytokines, chemokines, proteases | Accumulation of senescent cells and their SASP | Chronic inflammation (inflammaging), impaired stem cell function, senescence propagation |
| Extracellular Vesicles (EVs) | MicroRNAs, Proteins, Lipids | Altered cargo, reduced efficiency, pro-senescent EVs | Impaired immune modulation, reduced tissue repair, spread of detrimental signals |
| Gap Junctions | Connexins, Ions, Small Molecules | Reduced expression/function of connexins | Loss of tissue cohesion, dysregulated cell growth, impaired metabolic coupling |
| Neuroendocrine System | Hormones (e.g., sex hormones, GH), Neurotransmitters | Hormonal decline, reduced receptor sensitivity | Metabolic disorders, cognitive decline, immune dysfunction, sleep disturbances |
| Immune System | Cytokines, Chemokines, Immune Cells | Immunosenescence, chronic low-grade inflammation | Increased infection risk, autoimmune disorders, chronic inflammation (inflammaging) |
This widespread dysregulation appears to lead to:
- Tissue and Organ Dysfunction: Impaired communication can manifest as muscle wasting (sarcopenia), cognitive decline (neurodegeneration), cardiovascular disease, kidney dysfunction, and impaired wound healing.
- Reduced Regenerative Capacity: When stem cells receive faulty signals or are exposed to a hostile microenvironment (e.g., due to SASP), their ability to repair and replace damaged cells is compromised, leading to slower recovery from injury and chronic tissue degeneration.
- Increased Susceptibility to Disease: The chronic inflammation and cellular dysfunction stemming from communication breakdown are major risk factors for a host of age-related diseases, including type 2 diabetes, cancer, cardiovascular disease, and neurodegenerative disorders.
- Accelerated Biological Aging: Ultimately, the breakdown in intercellular communication contributes to the overall acceleration of biological aging, even if chronological age remains the same.
Targeting Intercellular Communication for Health and Longevity
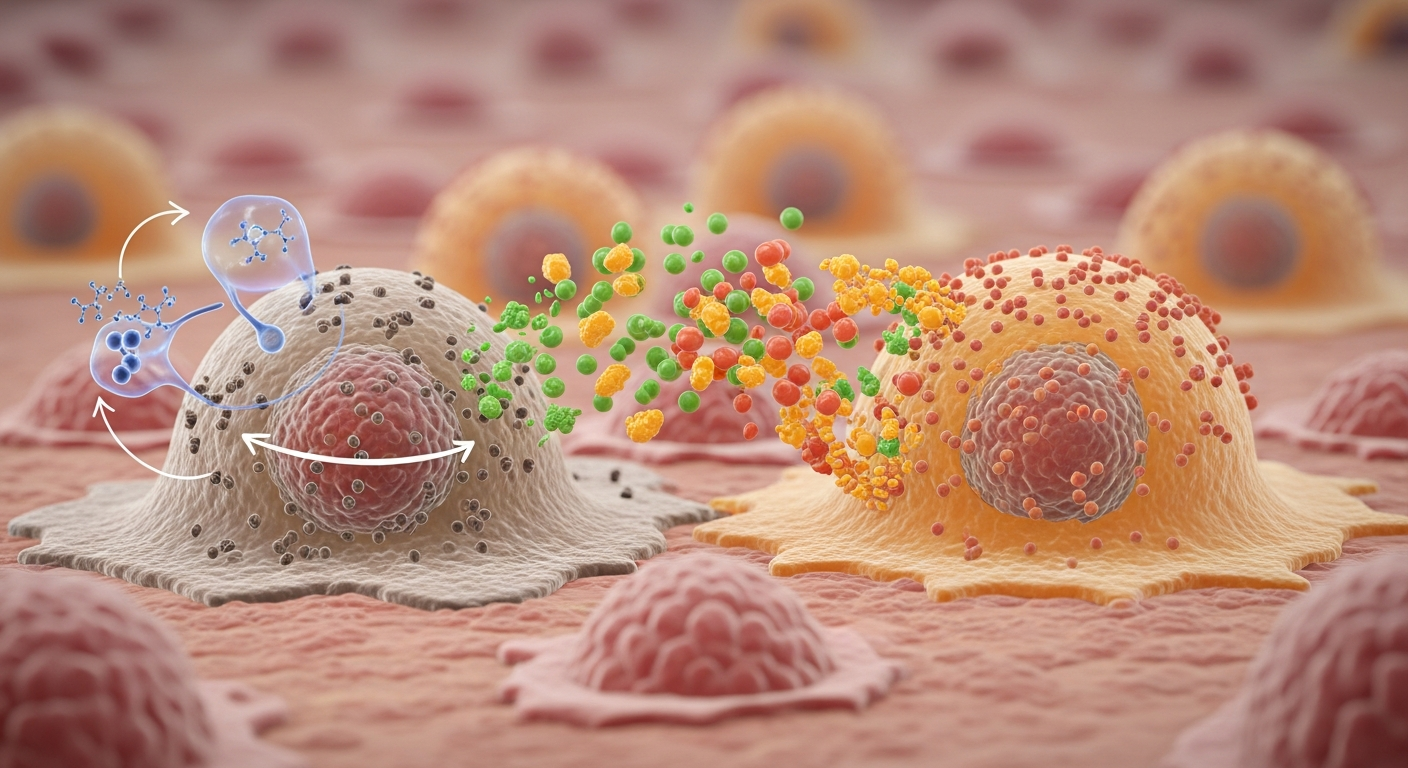
Given its fundamental role in aging, modulating intercellular communication represents a promising strategy for developing interventions to promote healthy aging and extend healthspan. Research is exploring both pharmacological and lifestyle approaches.
What Pharmacological Interventions Are Being Investigated?
Several classes of drugs are under investigation for their potential to restore youthful cellular communication.
Senolytics and Senomorphics
These compounds aim to address the detrimental effects of senescent cells and their SASP.
- Senolytics are drugs designed to selectively kill senescent cells. By clearing these “zombie cells,” senolytics aim to reduce the burden of SASP factors, thereby improving the microenvironment and restoring more effective communication between healthy cells. Examples include dasatinib combined with quercetin (D+Q) and fisetin (Kirkland & Tchkonia, 2017; PMID: 28629683). Early human trials show promise in reducing senescent cell burden and improving some age-related conditions.
- Senomorphics are agents that modify the SASP without necessarily killing the senescent cells, thereby reducing their harmful secretory output. This approach could also mitigate the disruptive effects of SASP on surrounding cells.
mTOR Inhibitors
Drugs like rapamycin and its analogs (rapalogs) inhibit the mTOR pathway. By dampening chronic mTOR activation, these compounds may promote autophagy, improve cellular metabolism, and potentially reduce the pro-aging signals associated with persistent mTOR activity. Rapamycin has shown consistent lifespan extension in various animal models, and clinical trials are exploring its potential benefits in humans for age-related conditions (Johnson et al., 2019; PMID: 31109918).
Metformin
This widely used anti-diabetic drug has garnered significant attention in longevity research. Metformin primarily acts by activating AMPK, which in turn modulates the insulin/IGF-1 and mTOR pathways, shifting cellular metabolism towards a more catabolic, energy-efficient state. By improving metabolic signaling and reducing chronic inflammation, metformin may help restore more balanced intercellular communication (Barzilai et al., 2016; PMID: 27173262). The TAME (Targeting Aging with Metformin) trial aims to investigate its potential to delay age-related diseases in non-diabetic individuals.
Hormonal Therapies (with caution)
While age-related hormonal decline is a clear example of communication breakdown, hormone replacement therapies (HRT) require careful consideration due to potential side effects. For instance, estrogen replacement therapy in post-menopausal women or testosterone replacement in men with hypogonadism can alleviate some symptoms of aging, but individualized risk-benefit assessments are crucial. The goal is to restore physiological levels and improve cellular responsiveness without inducing adverse effects.
What Lifestyle Interventions Can Improve Cellular Communication?
Beyond pharmacological agents, various lifestyle interventions are thought to positively influence intercellular communication, often by targeting the same pathways that drugs do, but through natural mechanisms.
Diet and Nutrition
- Calorie Restriction (CR): Reducing caloric intake without malnutrition has consistently extended lifespan and healthspan in diverse organisms. CR appears to improve cellular communication by enhancing nutrient sensing pathways (e.g., activating AMPK, inhibiting mTOR, modulating IIS), reducing inflammation, and promoting cellular repair mechanisms.
- Time-Restricted Eating/Intermittent Fasting: These eating patterns may mimic some benefits of CR by creating periods of nutrient scarcity that activate stress response pathways and improve metabolic flexibility, thereby optimizing cellular communication.
- Balanced, Nutrient-Dense Diet: Diets rich in antioxidants, anti-inflammatory compounds (e.g., polyphenols from fruits, vegetables, and whole grains), and healthy fats can help reduce oxidative stress and chronic inflammation, which are major disruptors of cellular communication. Avoiding ultra-processed foods, high sugar intake, and excessive saturated fats can mitigate metabolic dysfunction and inflammaging.
Regular Exercise
Physical activity is a powerful modulator of intercellular communication.
- Improved ECM Health: Exercise helps maintain the integrity and elasticity of the ECM, promoting better mechanotransduction and nutrient exchange.
- Reduced Inflammation: Regular exercise has anti-inflammatory effects, helping to counteract inflammaging and the detrimental effects of SASP.
- Metabolic Signaling: Exercise activates AMPK, improves insulin sensitivity, and modulates other growth factor signaling pathways, leading to more robust and balanced cellular communication.
- Hormonal Balance: Exercise can help maintain healthy levels of various hormones, including growth hormone and sex hormones, which support systemic communication.
Stress Management and Sleep
Chronic psychological stress can elevate cortisol levels and promote inflammation, disrupting cellular communication throughout the body. Techniques like mindfulness, meditation, and yoga can help manage stress responses. Quality sleep is also crucial for cellular repair and detoxification processes, which are essential for maintaining effective cellular communication networks. Sleep deprivation, conversely, can increase inflammation and metabolic dysregulation.
The Future of Intercellular Communication Research in Longevity
The study of intercellular communication breakdown in aging is a rapidly evolving field. Researchers are continually uncovering new signaling molecules, communication pathways, and the intricate ways they are affected by age. Advanced technologies, such as single-cell sequencing and sophisticated imaging techniques, are providing unprecedented insights into how individual cells within a tissue communicate and how these patterns change over time.
Moving forward, the focus is likely to be on:
- Precision Interventions: Tailoring treatments based on an individual’s unique cellular communication profile.
- Multi-target Approaches: Developing therapies that simultaneously address several aspects of communication breakdown (e.g., clearing senescent cells while also modulating metabolic pathways).
- Biomarkers of Communication Health: Identifying reliable biomarkers that can assess the health of intercellular communication networks and track the effectiveness of interventions.
By delving deeper into the “language” of our cells and understanding how it becomes garbled with age, we may unlock powerful new strategies to preserve health, prevent disease, and extend the period of robust well-being in later life.
Conclusion
Intercellular communication is the silent conductor orchestrating the symphony of life within our bodies. Its progressive breakdown with age, driven by dysregulated signaling pathways, a stiffened extracellular matrix, the accumulation of senescent cells and their disruptive SASP, altered extracellular vesicles, impaired direct cell contact, and neuroendocrine and immune system dysregulation, represents a fundamental hallmark of aging. This multifaceted deterioration contributes significantly to tissue dysfunction, impaired regeneration, and the increased incidence of age-related diseases.
However, recognizing this critical aspect of aging also illuminates promising avenues for intervention. Through both pharmacological agents
Frequently Asked Questions
What is intercellular communication?
How does aging affect cellular communication?
Can we improve intercellular communication to promote longevity?
Sources
- The hallmarks of aging(2013)
- Cellular senescence: a key determinant in health and disease(2019)
- Extracellular matrix and aging(2016)
- The secret life of extracellular vesicles in aging(2018)
- The insulin/IGF-1 signaling pathway and human longevity(2014)
- Inflammaging: a new immune-metabolic viewpoint for the oldest-old(2013)
- Targeting mTOR for health and longevity(2019)
- The role of gap junctions in aging(2010)
Stay Updated on Longevity Science
Weekly research digests. No spam, unsubscribe anytime.
Related Articles

Autophagy and Aging: Your Body's Cellular Cleanup System
Explore autophagy, your body's cellular cleanup system, and its critical role in aging and longevity. Research suggests modulating autophagy may support cellular health.
12 min read
Epigenetic Alterations: How Your Genes Age Without Changing
Understand how epigenetic alterations drive aging through DNA methylation changes, histone modifications, and chromatin remodeling -- and what research suggests.
14 min read
Genomic Instability and Aging: How DNA Damage Drives the Aging Process
Explore how genomic instability contributes to aging, the role of DNA damage and repair mechanisms, and emerging research on protecting genomic integrity.
15 min read